武汉华美生物工程有限公司CUSABIO®品牌商
15 年
手机商铺
- NaN
- 0
- 0
- 2
- 2
推荐产品
公司新闻/正文
CAR-T、ADC、癌症疫苗群雄逐鹿:MUC1靶点研发全景解析
530 人阅读发布时间:2026-03-06 11:52
1. MUC1的结构背景与生理功能
黏蛋白家族包括跨膜型(如MUC1、MUC4、MUC16)、分泌型及可溶性等多种成员。其中,MUC1是该家族中特征最明确的跨膜黏蛋白,定位于上皮细胞顶面,参与上皮保护、修复、细胞黏附及免疫应答等多种生理过程。
结构上,MUC1是由非共价结合的N端(MUC1-N)和C端(MUC1-C)两个亚基组成的异二聚体2121。MUC1-N位于胞外,包含数量可变的20氨基酸串联重复序列(VNTR),该区域富含O-糖基化位点。高度糖基化的MUC1-N向细胞外延伸,在黏膜表面形成物理屏障,保护上皮细胞免受外界损伤。MUC1-C则由胞外段、跨膜段和胞质尾区组成,是信号转导的关键部分。在SEA结构域介导下,MUC1可发生自蛋白水解切割;当接收到炎症信号(如IFN-γ、TNF-α)时,MUC1-N被释放,进而激活MUC1-C,启动下游信号通路 [27]。
在正常组织中,MUC1呈严格的极性表达,仅位于细胞顶端膜面,维持上皮屏障完整性并避免异常信号激活。在感染或炎症环境下,MUC1可通过糖链末端唾液酸结构与病毒结合(如限制甲流感染),同时调节炎症强度,防止免疫损伤。因此,生理状态下MUC1兼具屏障功能与免疫调节作用。
然而,在慢性炎症、缺氧或遗传易感背景下,MUC1极性表达丧失、全细胞表面异常过表达,并由“物理防御者”转变为“信号枢纽分子”,成为多种恶性疾病的关键驱动因素。
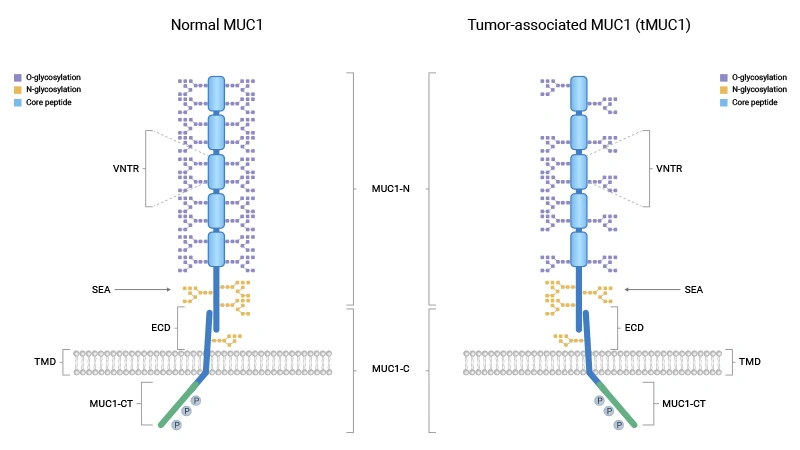
图. MUC1结构在生理状态(正常MUC1)与癌症状态(tMUC1)下的示意图 [27]
2. 遗传易感性与表观遗传调控
MUC1的异常表达与遗传背景密切相关。全基因组关联研究已确认1q22区域为胃癌重要易感位点,其中功能性单核苷酸多态性rs4072037受到广泛关注 [1,2]。多项荟萃分析显示,G等位基因显著降低胃癌风险,其效应值(OR)通常位于0.70–0.79之间 [3-5]。在东亚人群中,该位点的人群归因危险度较高 [6,7],提示其具有流行病学意义。该多态性不仅影响胃癌总体风险,还与弥漫型胃癌的预后密切相关 [10],并在癌前病变阶段即表现出关联 [11]。
在表观遗传层面,MUC1启动子甲基化状态是调控其表达的关键因素。在胰腺导管腺癌中,启动子低甲基化与mRNA高表达显著相关,并提示不良预后 [15]。类似的表观遗传异常也在肺腺癌中得到验证 [16]。此外,功能性变异如rs4072037及rs12411216还与代谢及肾功能指标相关 [17],体现出MUC1的多系统多效性。
总体而言,MUC1的遗传与表观遗传背景为其异常表达提供了分子基础,但其致病效应仍需通过下游信号网络得以实现。
3. 异常糖基化与关键信号通路机制
MUC1在肿瘤中的功能转化,核心依赖于糖基化异常与胞质尾区信号激活。肿瘤细胞中C1GALT1及Cosmc表达异常,导致Tn和sTn抗原暴露 [19]。这种糖链截断改变了MUC1构象,并暴露出新的配体结合位点。
Galectin-3可结合异常糖基化MUC1,诱导MUC1-C磷酸化并激活ERK1/2与Akt信号通路 [21]。与此同时,YBX1在转录层面增强MUC1表达 [20]。在空间调控上,ADAM10/17及γ-分泌酶介导MUC1-C水解释放并进入细胞核 [26],与β-Catenin形成复合体,调控EMT及干性相关基因表达 [9]。此外,MUC1-C通过JAK/STAT3与NF-κB信号增强PD-L1表达及免疫抑制环境形成 [18]。
因此,MUC1从膜结构分子转变为核内转录调控协同因子,是其驱动恶性转化的关键步骤。
4. MUC1在疾病中的作用与临床意义
MUC1异常表达贯穿肿瘤发生、进展与免疫逃逸全过程,其在多种实体瘤中具有明确的病理与临床意义。
在胃癌中,rs4072037多态性不仅影响总体发病风险,还与弥漫型胃癌的复发及疾病相关死亡密切相关 [10]。研究显示,MUC1表达异常在重度肠上皮化生及非典型增生阶段即已出现 [11],提示其在炎症向癌变转化过程中具有早期驱动作用。与幽门螺杆菌感染的协同效应进一步强化了这一连续谱模型 [12,13]。遗传易感性与慢性感染的叠加,使MUC1成为连接炎症与胃癌发生的重要分子桥梁。
在胰腺导管腺癌(PDAC)中,MUC1被识别为蛋白互作网络核心基因之一 [22]。组织病理学研究显示,其在癌组织中的表达显著高于正常导管组织 [25]。基于胰液DNA甲基化的检测方法可在较高特异性与灵敏度下区分PDAC与IPMN [23],显示出其在早期鉴别诊断中的潜在应用价值。值得注意的是,MUC1不仅是诊断标志物,其胞质尾区通过持续激活PI3K/Akt信号维持肿瘤细胞生存优势,提示其在治疗靶向中的重要地位。
在乳腺癌中,MUC1过表达被证实为不良预后的独立预测因子 [8]。其异常糖基化形式增强与E-selectin及ICAM-1的相互作用 [24],促进循环肿瘤细胞在血管内皮上的“滚动”与黏附,从而加速远处转移。此外,MUC1通过增强PD-L1表达及NF-κB活化构建免疫抑制微环境,使肿瘤细胞逃避免疫清除。
在非小细胞肺癌中,EIF4G1驱动的MUC1高表达与PD-L1水平显著相关 [14],并通过JAK/STAT3通路增强免疫逃逸能力。其在外泌体中的高度富集 [40] 进一步提示其在液体活检中的应用前景。
除实体瘤外,MUC1功能变异还与心血管代谢指标及肾功能参数相关 [17],表明其在系统性炎症与代谢调控中也可能发挥作用。虽然其在非肿瘤疾病中的机制尚未完全阐明,但多效性特征提示其可能参与慢性炎症与组织纤维化进程。
总体来看,MUC1在不同疾病中的作用呈现出共同特征:表达极性丧失、糖基化异常、核转位增强以及免疫调控失衡。这些机制构成其在多癌种中的“共享致病框架”。
5. MUC1靶向药物最近研究进展
目前,MUC1靶向药物研发已进入以免疫治疗和精准偶联技术为主导的多元化临床阶段。从药物类型来看,细胞疗法(CAR-T)和抗体偶联药物(ADC)构成了研发双主线,进展最快的候选药物多处于临床II期,适应症主要集中于前列腺癌、胰腺癌、非小细胞肺癌等晚期实体瘤。值得关注的是,CAR-T疗法研发尤为活跃,以广州安捷、深圳免疫基因治疗研究院、上海斯丹赛、广东昭泰等为代表的国内生物技术公司均有管线进入临床,显示出中国在MUC1细胞治疗领域的深度布局。部分在研管线整理如下表:
| 药物 | 作用机制 | 药物类型 | 在研适应症 | 在研机构 | 最高研发阶段 |
|---|---|---|---|---|---|
| Anti-MUC1 CAR T cell(Guangzhou Anjie Biomedical Technology Co., Ltd.) | MUC1调节剂 | T淋巴细胞替代物 | CAR-T | 前列腺癌 | 广州安捷生物医学技术有限公司 | 临床2期 |
| GO-2032c | MUC1抑制剂 | 合成多肽 | 急性髓性白血病 | Dana-Farber Cancer Institute, Inc. | Genus Oncology LLC | 临床2期 |
| Anti-MUC1 monoclonal antibody(AltaRex Corp.) | MUC1抑制剂 | 单克隆抗体 | 胰腺癌 | HYULIM A-TECH Co., Ltd. | ViRexx Medical Corp. | 深圳市海普瑞药业集团股份有限公司 | 临床2期 |
| Cancer vaccine(Therion Biologics Corp.) | CEACAM5拮抗剂 | MUC1抑制剂 | 免疫刺激剂 | 重组载体疫苗 | 治疗性疫苗 | 小肠癌 | 结直肠癌 | 非小细胞肺癌 | 前列腺癌等 | National Cancer Institute | Bavarian Nordic, Inc. | 临床2期 |
| Adenovirus 5 CEA/MUC1/Brachyury Vaccine(National Cancer Institute) | CEA抑制剂 | MUC1抑制剂 | TBXT抑制剂 | 免疫刺激剂 | 重组载体疫苗 | 治疗性疫苗 | 遗传性非息肉病性结直肠癌 | National Cancer Institute | 临床2期 |
| CV-9202 | 5T4抑制剂 | MAGEC1拮抗剂 | MAGEC2拮抗剂 | MUC1抑制剂 | NY-ESO-1抑制剂 | survivin抑制剂 | 治疗性疫苗 | mRNA疫苗 | 非小细胞肺癌 | Ludwig Institute for Cancer Research Ltd. | C.H. Boehringer Sohn AG & Co. KG | 临床2期 |
| 吉西他滨脂质体 | DNA polymerase抑制剂 | MUC1抑制剂 | 小分子化药 | 脂质体药物 | 晚期非小细胞肺癌 | 晚期恶性实体瘤 | 晚期尿路上皮癌等 | FUJIFILM Pharmaceuticals USA, Inc. | FUJIFILM Holdings America Corp. | 临床2期 |
| Cancer vaccine-MUC 1(Vaxil Biotherapeutics) | MUC1抑制剂 | 治疗性疫苗 | 乳腺癌 | 多发性骨髓瘤 | Vaxil Biotherapeutics Ltd. | Hadassah Foundation | 临床2期 |
| MUC1-Poly-ICLC | MUC1调节剂 | 治疗性疫苗 | 非小细胞肺癌 | University of Pittsburgh Medical Center | 临床2期 |
| Monoclonal antibody HMFG1(Cancer Research UK) | MUC1抑制剂 | 免疫刺激剂 | 单克隆抗体 | 局部晚期乳腺癌 | 转移性乳腺癌 | Antisoma Research Ltd. | 临床2期 |
(数据截止到2026年2月27日,来源于synapse)
6. MUC1相关研究工具
针对MUC1从基础机制到临床转化的广阔研究前景,华美生物(CUSABIO)提供覆盖上述研究需求的完整工具产品线,包括重组蛋白、抗体以及ELISA试剂盒,助力您在遗传调控、信号通路、药物筛选及液体活检等各环节的研究。
● MUC1重组蛋白
Recombinant Human Mucin-1 (MUC1), partial; Code: CSB-MP015215HU2
Recombinant Human Mucin-1(MUC1),partial,Biotinylated;
CSB-MP015215HU3-B

Recombinant Mouse Mucin-1 (Muc1), partial;
CSB-EP312543MO
● MUC1抗体
MUC1 Recombinant Monoclonal Antibody; CSB-RA049212A0HU
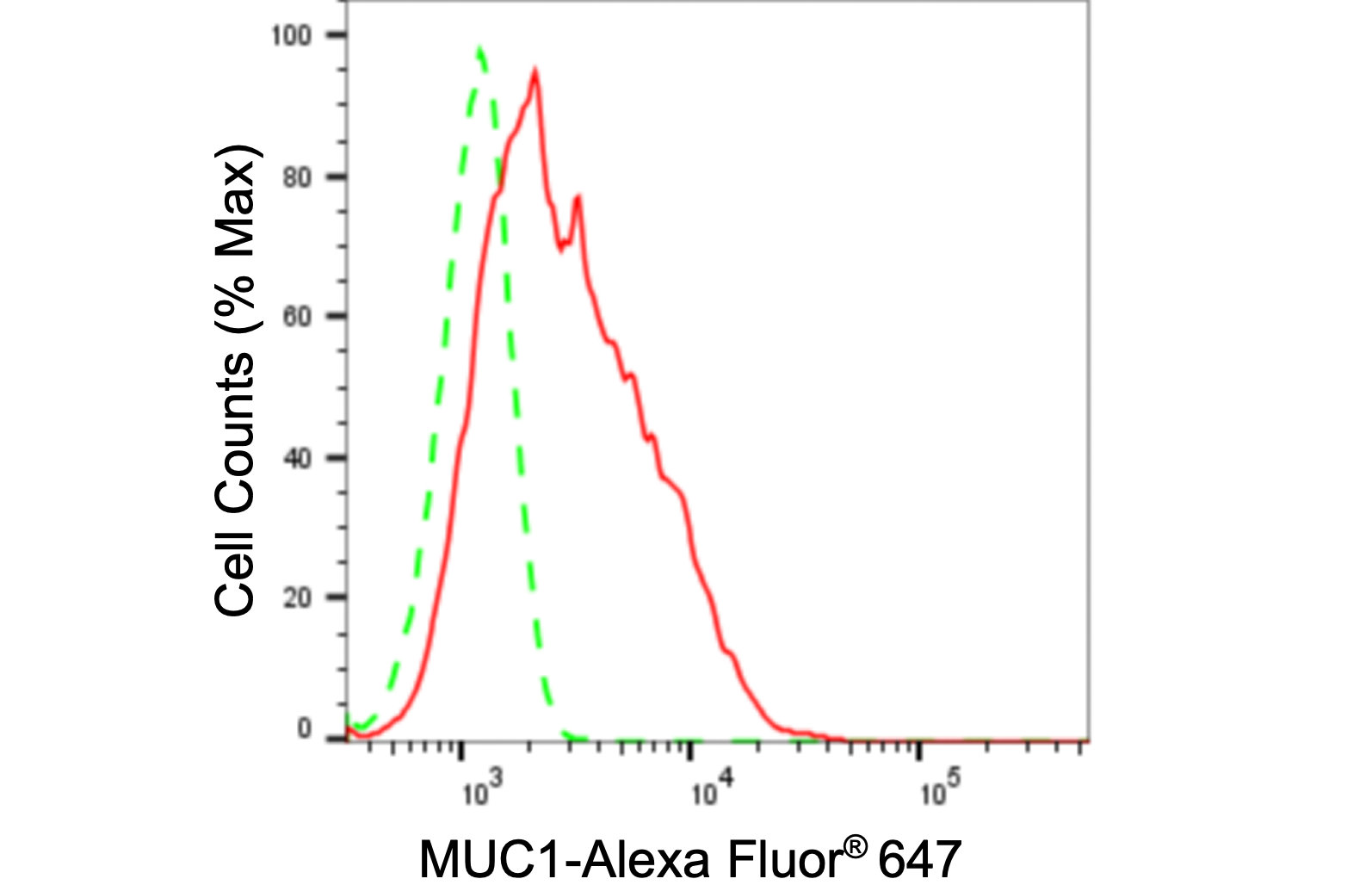
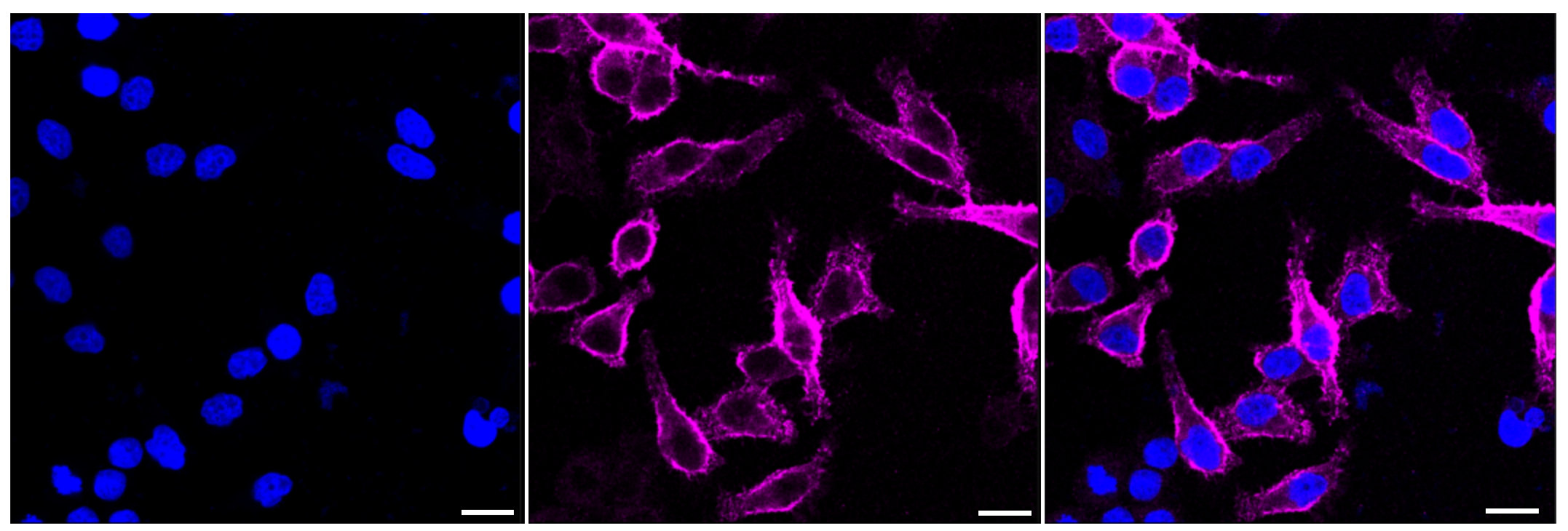
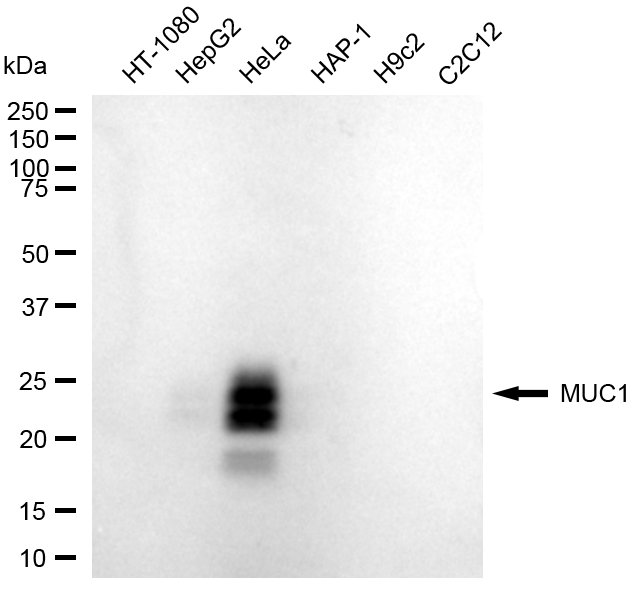
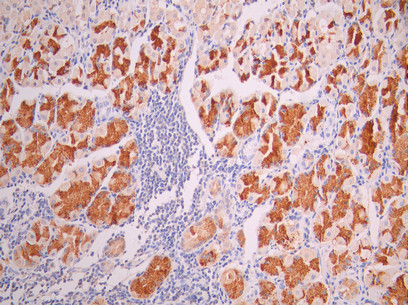
MUC1 Recombinant Monoclonal Antibody; CSB-RA049212A0HU
● MUC1 ELISA试剂盒
Human mammary carcinoma marker CA15-3 (CA15-3) ELISA kit; CSB-E04772h
[1] Tomomitsu Tahara, Sayumi Tahara, Noriyuki Horiguchi, Takema Kato, Yasuko Shinkai, Masaaki Okubo, Tsuyoshi Terada, Dai Yoshida, Kohei Funasaka, Mitsuo Nagasaka, Yoshihito Nakagawa, Hiroki Kurahashi, Tomoyuki Shibata, Tetsuya Tsukamoto, Naoki Ohmiya.(2019). Prostate Stem Cell Antigen Gene Polymorphism Is Associated withH. pylori–related Promoter DNA Methylation in Nonneoplastic Gastric Epithelium.
[2] Norihisa Saeki, Hiromi Sakamoto, Teruhiko Yoshida.(2014). Mucin 1 Gene (MUC1) and Gastric-Cancer Susceptibility.
[3] Peixi Liu, Mingxi Zeng.(2020). Role ofMUC1rs4072037 polymorphism in gastric cancer: a meta-analysis.
[4] Xi Gu, Jiaxi Feng, Liyang Liu, Ming Lü, Xiang Ma, Yang Cao, Baolin Wang, Qinghong Zhao.(2018). Association of MUC1 rs4072037 Functional Polymorphism and Cancer Risk: Evidence from 12551 Cases and 13436 Controls.
[5] Fujiao Duan, Chunhua Song, Liping Dai, Shuli Cui, Xiaoqin Zhang, Xia Zhao.(2014). The Effect of MUC1 rs4072037 Functional Polymorphism on Cancer Susceptibility: Evidence from Published Studies.
[6] Fujiao Duan, Chunhua Song, Jiachen Shi, Yanchun Wang, Hua Ye, Liping Dai, Jianying Zhang, Kaijuan Wang.(2021). Identification and epidemiological evaluation of gastric cancer risk factors: based on a field synopsis and meta-analysis in Chinese population.
[7] Hongwei Sun, Xiaoli Wu, Fang Wu, Ying Li, Zhengping Yu, Xiangrong Chen, Yunzhi Chen, Wenjun Yang.(2015). Associations of Genetic Variants in the PSCA, MUC1 and PLCE1 Genes with Stomach Cancer Susceptibility in a Chinese Population.
[8] Xuan Jing, Hongping Liang, Chonghua Hao, Xiaojuan Yang, Xiangrong Cui.(2018). Overexpression of MUC1 predicts poor prognosis in patients with breast cancer.
[9] Andreia Mota Sousa, Margarida Rei, Rita Freitas, Sara Ricardo, Thomas C. Caffrey, Leonor David, Raquel Almeida, Michael A. Hollingsworth, Filipe Santos‐Silva.(2016). Effect of MUC1/β-catenin interaction on the tumorigenic capacity of pancreatic CD133+ cells.
[10] Beom Su Kim, Inchul Lee, Jeong Hwan Yook, Kyuyoung Song, Byung-Sik Kim.(2020). Association between theMUC1rs4072037 Polymorphism and Risk of Gastric Cancer and Clinical Outcomes.
[11] Meng Cai, Shuyang Dai, Wanqing Chen, Changfa Xia, Lingeng Lu, Shuguang Dai, Jun Qi, Minjie Wang, Meilin Wang, Lanping Zhou, Fuhua Lei, Tingting Zuo, Hongmei Zeng, Xiaohang Zhao.(2017). Environmental factors, seven GWAS‐identified susceptibility loci, and risk of gastric cancer and its precursors in a Chinese population.
[12] Luca Giraldi, Maria Benedetta Michelazzo, Dario Arzani, Roberto Persiani, Roberta Pastorino, Stefania Boccia.(2017). MUC1, MUC5AC, and MUC6 polymorphisms, Helicobacter pylori infection, and gastric cancer: a systematic review and meta-analysis.
[13] Miao Li, Liu Huang, Hong Qiu, Qiang Fu, Wen Li, Qianqian Yu, Li Sun, Lihong Zhang, Guangyuan Hu, Junbo Hu, Xianglin Yuan.(2013). Helicobacter pylori Infection Synergizes with Three Inflammation-Related Genetic Variants in the GWASs to Increase Risk of Gastric Cancer in a Chinese Population.
[14] Luis Del Valle, Lu Dai, Hui‐Yi Lin, Zhen Lin, Jungang Chen, Steven R. Post, Zhiqiang Qin.(2021). Role of EIF4G1 network in non‐small cell lung cancers (NSCLC) cell survival and disease progression.
[15] Seiya Yokoyama, Michiyo Higashi, Sho Kitamoto, Monika Oeldorf, Uwe Knippschild, Marko Kornmann, Kosei Maemura, Hiroshi Kurahara, Edwin Wiest, Tomofumi Hamada, Ikumi Kitazono, Yuko Goto‐Koshino, Takashi Tasaki, Tsubasa Hiraki, Kazuhito Hatanaka, Yuko Mataki, Hiroki Taguchi, Shinichi Hashimoto, Surinder K. Batra, Akihide Tanimoto, Suguru Yonezawa, Michael A. Hollingsworth.(2016). Aberrant methylation ofMUC1andMUC4promoters are potential prognostic biomarkers for pancreatic ductal adenocarcinomas.
[16] Xiaocong Wang, Yanmei Li, Huihua Hu, Fangzheng Zhou, Jie Chen, Dongsheng Zhang.(2020). Comprehensive analysis of gene expression and DNA methylation data identifies potential biomarkers and functional epigenetic modules for lung adenocarcinoma.
[17] Ming‐Sheng Teng, Semon Wu, Lung‐An Hsu, Hsin‐Hua Chou, Yu‐Lin Ko.(2021). Pleiotropic Effects of Functional MUC1 Variants on Cardiometabolic, Renal, and Hematological Traits in the Taiwanese Population.
[18] Saumya Mishra, Patrícia Mendonça, Sukhmandeep Kaur, Karam F. A. Soliman.(2025). Silibinin Anticancer Effects Through the Modulation of the Tumor Immune Microenvironment in Triple-Negative Breast Cancer.
[19] Hong Tian, Jiali Yu, Xiaoli Chu, Qi Guan, Juan Liu, Ying Liu.(2024). Unraveling the role of C1GALT1 in abnormal glycosylation and colorectal cancer progression.
[20] Qiang Xie, Shilei Zhao, Wenzhi Liu, Yanwei Cui, Fengzhou Li, Zhuoshi Li, Tao Guo, Wendan Yu, Wei Guo, Wuguo Deng, Chundong Gu.(2021). YBX1 Enhances Metastasis and Stemness by Transcriptionally Regulating MUC1 in Lung Adenocarcinoma.
[21] Yugo Mori, Kaoru Akita, Masakazu Yashiro, Tetsuji Sawada, Kosei Hirakawa, Takeomi Murata, Hiroshi Nakada.(2015). Binding of Galectin-3, a β-Galactoside-binding Lectin, to MUC1 Protein Enhances Phosphorylation of Extracellular Signal-regulated Kinase 1/2 (ERK1/2) and Akt, Promoting Tumor Cell Malignancy.
[22] Yan Li, Yuzhang Zhu, Guiping Dai, Dongjuan Wu, Zhenzhen Gao, Lei Zhang, Yaohua Fan.(2019). Screening and validating the core biomarkers in patients with pancreatic ductal adenocarcinoma.
[23] Seiya Yokoyama, Sho Kitamoto, Michiyo Higashi, Yuko Goto‐Koshino, Taro Hara, Dai Ikebe, Taketo Yamaguchi, Yoshifumi Arisaka, Toru Niihara, Hiroto Nishimata, Sadao Tanaka, Kyoichi Takaori, Surinder K. Batra, Suguru Yonezawa.(2014). Diagnosis of Pancreatic Neoplasms Using a Novel Method of DNA Methylation Analysis of Mucin Expression in Pancreatic Juice.
[24] Yue Geng, Kimberly Yeh, Tait Takatani, Michael R. King.(2012). Three to Tango: MUC1 as a Ligand for Both E-Selectin and ICAM-1 in the Breast Cancer Metastatic Cascade.
[25] Alberto Nicoletti, Federica Vitale, Giuseppe Quero, Mattia Paratore, Claudio Fiorillo, Marcantonio Negri, Angela Carlino, Frediano Inzani, Antonio Gasbarrini, Sergio Alfieri, Lorenzo Zileri Dal Verme.(2023). Immunohistochemical Evaluation of the Expression of Specific Membrane Antigens in Patients with Pancreatic Ductal Adenocarcinoma.
[26] Audrey Bouillez, Viviane Gnemmi, Kelly Gaudelot, Brigitte Hémon, Bélinda Ringot, Nicolas Pottier, François Glowacki, Caroline Butruille, Christelle Cauffiez, Malika Hamdane, Nicolas Sergeant, Isabelle Van Seuningen, Xavier Leroy, Sébastien Aubert, Michaël Perrais.(2014). MUC1-C nuclear localization drives invasiveness of renal cancer cells through a sheddase/gamma secretase dependent pathway.
[27] Rosalia Dieli, Rosa Lioy, Fabiana Crispo, Nicoletta Cascelli, Mara Martinelli Donatella Telesca, Maria Rita Milella, Marco Colella, Simona Loperte, Rosa Lerose. Biomolecules.(2025).The Oncoprotein Mucin 1 in Pancreatic Cancer Onset and Progression: Potential Clinical Implications.



