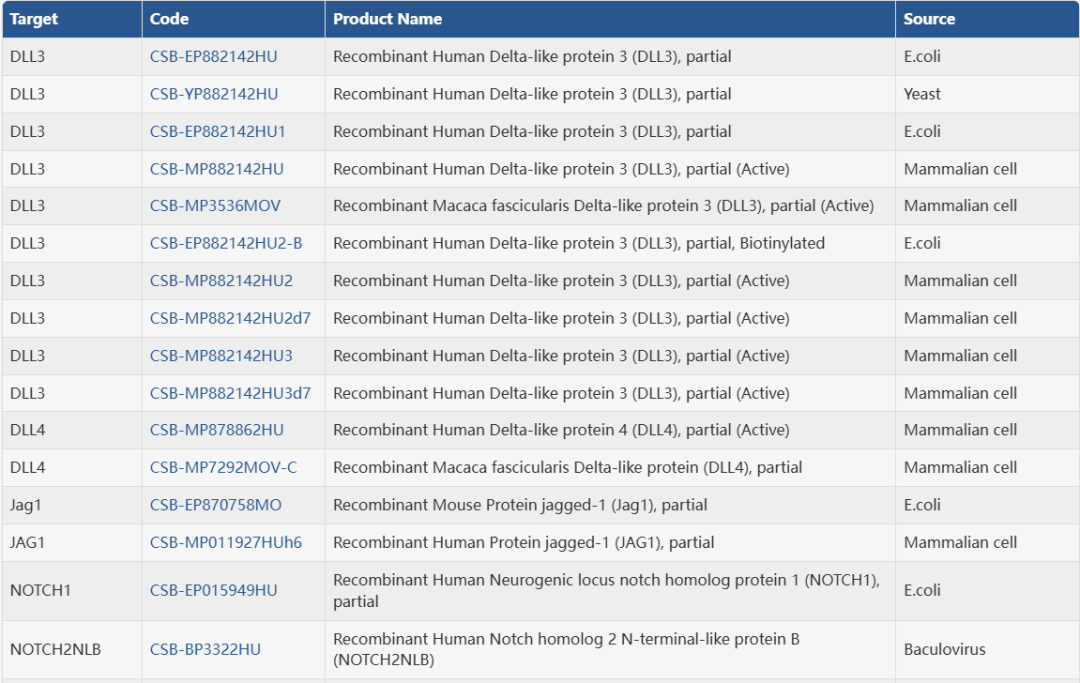
武汉华美生物工程有限公司CUSABIO®品牌商
15 年
手机商铺
- NaN
- 0
- 0
- 2
- 2
推荐产品
公司新闻/正文
Notch信号通路研究必备:从结构到疾病,一文读懂
176 人阅读发布时间:2026-03-20 11:43
Notch信号通路在生物体内扮演着至关重要的角色,不仅调控胚胎发育和成体组织稳态,更在多种癌症的发生、发展中发挥双重作用——既可作为肿瘤抑制因子,也能驱动癌细胞增殖、血管生成及免疫逃逸。本专题页面系统梳理Notch1–4受体与DLL/JAG配体的核心知识,助您快速定位高品质重组蛋白、抗体及ELISA试剂盒,推进肿瘤及其他疾病领域的Notch相关研究。
Notch信号通路是进化上高度保守的细胞间通讯系统,最早因果蝇翅膀“缺刻”表型而被发现。在哺乳动物中,该通路由4种跨膜受体(Notch1–4)和5种经典配体(DLL1、DLL3、DLL4、JAG1、JAG2)组成,仅在相邻细胞直接接触时被激活,精准调控细胞命运决定、分化、增殖及组织稳态。
所有Notch受体均为I型跨膜蛋白,在高尔基体中经furin酶切后以异二聚体形式定位于细胞膜;配体同样为跨膜蛋白,表达于邻近细胞表面。当配体与受体结合后,触发两次蛋白水解切割(ADAM介导的S2切割和γ-secretase介导的S3切割),释放具有转录活性的Notch胞内结构域(NICD)。NICD进入细胞核,与RBP-J等因子形成转录复合物,激活Hes/Hey等靶基因表达,从而传递信号。
Notch信号通路是进化上高度保守的细胞间通讯系统,最早因果蝇翅膀“缺刻”表型而被发现。在哺乳动物中,该通路由4种跨膜受体(Notch1–4)和5种经典配体(DLL1、DLL3、DLL4、JAG1、JAG2)组成,仅在相邻细胞直接接触时被激活,精准调控细胞命运决定、分化、增殖及组织稳态。
所有Notch受体均为I型跨膜蛋白,在高尔基体中经furin酶切后以异二聚体形式定位于细胞膜;配体同样为跨膜蛋白,表达于邻近细胞表面。当配体与受体结合后,触发两次蛋白水解切割(ADAM介导的S2切割和γ-secretase介导的S3切割),释放具有转录活性的Notch胞内结构域(NICD)。NICD进入细胞核,与RBP-J等因子形成转录复合物,激活Hes/Hey等靶基因表达,从而传递信号。
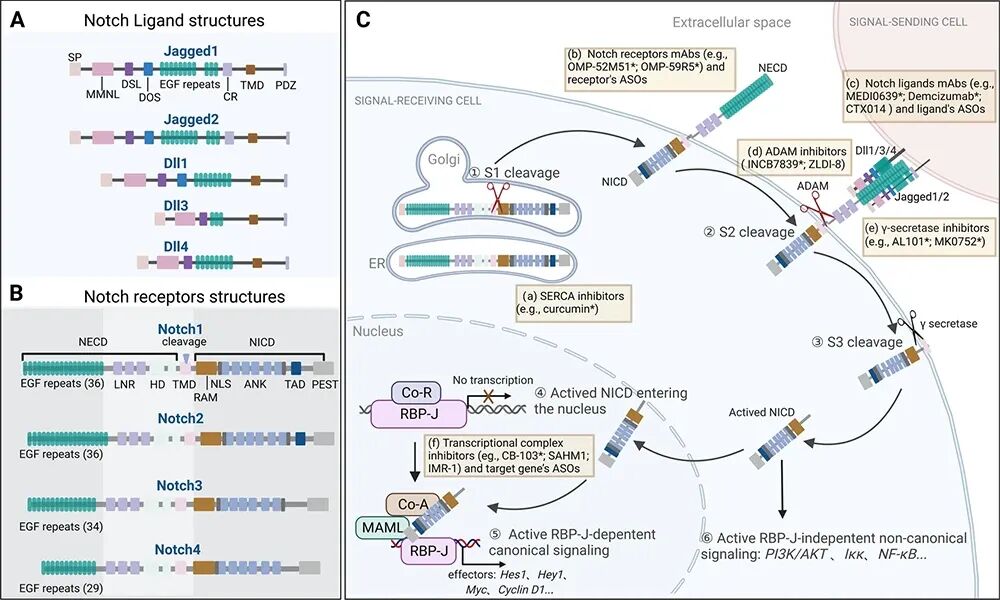
图 NOTCH信号通路 [1]
(A. 配体结构特征;B. 受体结构特征;C. 信号接收与转导过程。)
尽管通路机制高度相似,不同Notch受体与配体在组织分布、结合偏好及生物学功能上存在显著差异——例如DLL4-Notch1主导血管生成,JAG1-Notch2参与肾脏发育,而DLL3则在小细胞肺癌中异常高表达。这些特异性决定了研究中靶点选择与试剂匹配的重要性。
Notch信号通路的功能多样性,源于其受体与配体在不同组织和发育阶段的差异表达,以及由此产生的信号输出特异性。下表汇总了四种Notch受体与五种经典配体的表达特征、核心生物学功能及相关的疾病背景,为您提供快速参考。
● Notch受体家族(Notch1-4)
|
|
|
|
|
|---|---|---|---|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
● Notch配体家族(DLL/JAG)
|
|
|
|
|
|---|---|---|---|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
DLL3在超过80%的小细胞肺癌(SCLC)中高表达,且几乎不在正常组织中出现,使其成为理想的肿瘤特异性抗原。
机制特点:作为非典型配体,DLL3定位于高尔基体和细胞膜,通过反式抑制调控Notch信号,促进神经内分泌表型。
临床进展:
-
ovalpituzumab tesirine(Rova-T):靶向DLL3的抗体偶联药物(ADC),虽在III期未达主要终点,但验证了靶点可行性; -
Tarlatamab(BiTE双特异性T细胞衔接器):2024年获FDA突破性疗法认定,用于复发SCLC,客观缓解率超40%; -
目前,多款靶向DLL3的双特异性抗体、ADC药物和CAR-T疗法已进入临床研究阶段,展现出显著治疗潜力。
深入阅读:DLL3靶点新突破:再鼎医药公布ZL-1310早期临床数据!查看所有DLL3相关产品
DLL4 在血管新生过程中发挥负调控作用——通过激活内皮细胞上的 Notch1 受体,抑制过度出芽,维持血管网络的有序性。然而,在多种实体瘤(如乳腺癌、结直肠癌、肝癌)中,DLL4 异常高表达,导致肿瘤血管结构紊乱、灌注不足,反而促进缺氧、侵袭和免疫逃逸。
机制特点:靶向 DLL4 可诱导“非功能性血管增生”,虽增加血管密度,却破坏其成熟性,加剧肿瘤内部缺氧并抑制生长。更值得关注的是,DLL4 抑制可重塑肿瘤免疫微环境,增强 T 细胞浸润,与免疫检查点抑制剂(如抗 PD-1)产生协同效应。
临床进展:
-
目前进展最快的 DLL4 靶向药物是 Navicixizumab(OncoMed),一种 DLL4/VEGF 双抗,已进入铂耐药卵巢癌的 III 期临床试验。
深入阅读:DLL4:血管生成调控的关键靶点
查看所有DLL4相关产品Notch 信号通路在发育、稳态及肿瘤等关键生物学过程中发挥核心调控作用,其深入研究离不开高特异性、高可靠性的实验工具。华美生物提供覆盖 Notch 全家族受体与配体的抗体、重组蛋白及 ELISA 试剂盒,助力您的科研精准高效推进。
● 部分数据展示
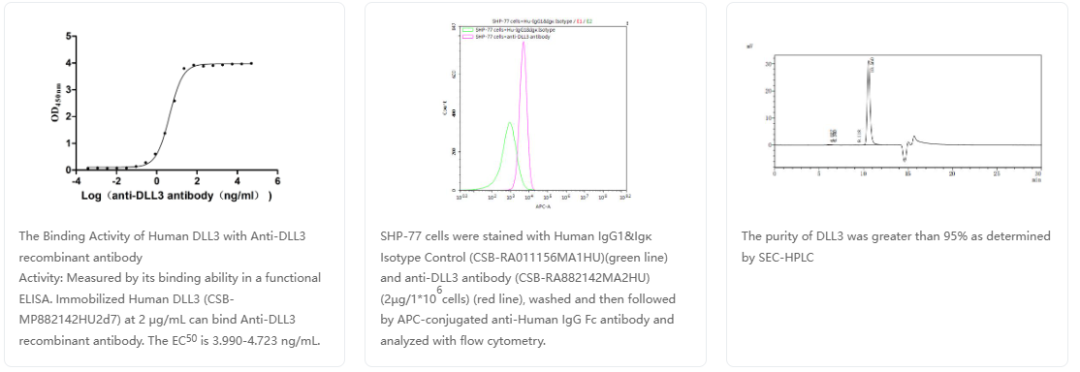
DLL3重组抗体
货号:CSB-RA882142MA2HU
人DLL3重组蛋白
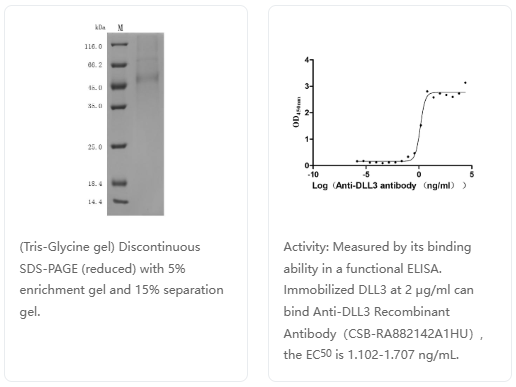
货号:CSB-MP882142HU
人DLL3 ELISA试剂盒
货号:CSB-EL006948HU
● 产品目录
重组蛋白
抗体
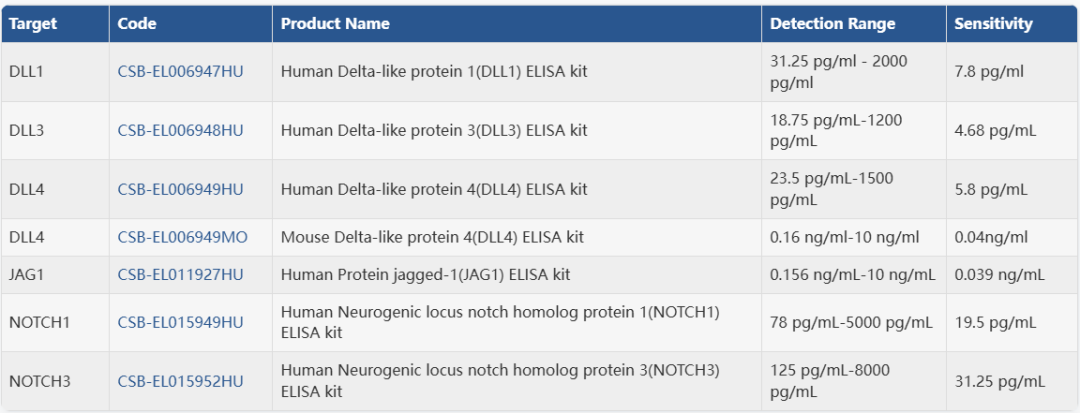
ELISA试剂盒
参考文献:
[1] Li X,Yan X,Wang Y, et al. The Notch signaling pathway: a potential target for cancer immunotherapy. J Hematol Oncol. 2023;16 (1):45.
[2] Herranz D,Ambesi-Impiombato A,Palomero T, et al. A NOTCH1-driven MYC enhancer promotes T cell development, transformation and acute lymphoblastic leukemia. Nat Med. 2014;20 (10):1130-7.
[3] Bertulfo K,Perez-Duran P,Miller H, et al. Therapeutic targeting of the NOTCH1 and neddylation pathways in T cell acute lymphoblastic leukemia. Proc Natl Acad Sci U S A. 2025;122 (14):e2426742122.
[4] Lei JH,Xu J,Lyu X, et al. NOTCH1 activation compensates BRCA1 deficiency and promotes triple-negative breast cancer formation. Nat Commun. 2020;11 (1):3256.
[5] Qiu H,Zmina PM,Huang AY, et al. Inhibiting Notch1 enhances immunotherapy efficacy in melanoma by preventing Notch1 dependent immune suppressive properties. Cancer Lett. 2018;434:144-151.
[6] Lindblad KE,Donne R,Liebling I, et al. NOTCH1 Drives Sexually Dimorphic Immune Responses in Hepatocellular Carcinoma. Cancer Discov. 2025;15 (3):495-510.
[7] Lu Y,Cao Y,Guo X, et al. Notch-Targeted Therapeutic in Colorectal Cancer by Notch1 Attenuation Via Tumor Microenvironment-Responsive Cascade DNA Delivery. Adv Healthc Mater. 2024;13 (22):e2400797.
[8] Hubmann R,Schwarzmeier JD,Shehata M, et al. Notch2 is involved in the overexpression of CD23 in B-cell chronic lymphocytic leukemia. Blood. 2002;99 (10):3742-7.
[9] Nayak R,Booker MA,Wang T, et al. Loss of NOTCH2 creates a TRIM28-dependent vulnerability in small cell lung cancer. Dev Cell. 2025;60 (24):3462-3479.e13.
[10] Xu J,Xu W,Yang X, et al. LncRNA MIR99AHG mediated by FOXA1 modulates NOTCH2/Notch signaling pathway to accelerate pancreatic cancer through sponging miR-3129-5p and recruiting ELAVL1. Cancer Cell Int. 2021;21 (1):674.
[11] Lee GH,Yoo KC,An Y, et al. FYN promotes mesenchymal phenotypes of basal type breast cancer cells through STAT5/NOTCH2 signaling node. Oncogene. 2018;37 (14):1857-1868.
[12] Heidari P,Taghizadeh M,Vakili O. Signaling pathways and molecular mechanisms involved in the onset and progression of cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL); a focus on Notch3 signaling. J Headache Pain. 2025;26 (1):96.
[13] Cardinale CJ,Li D,Tian L, et al. Association of a rare NOTCH4 coding variant with systemic sclerosis: a family-based whole exome sequencing study. BMC Musculoskelet Disord. 2016;17 (1):462.
[14] Bonyadi Rad E,Hammerlindl H,Wels C, et al. Notch4 Signaling Induces a Mesenchymal-Epithelial-like Transition in Melanoma Cells to Suppress Malignant Behaviors. Cancer Res. 2016;76 (7):1690-7.
[15] Gramantieri L,Giovannini C,Lanzi A, et al. Aberrant Notch3 and Notch4 expression in human hepatocellular carcinoma. Liver Int. 2007;27 (7):997-1007.
[16] Harb H,Chatila TA. Recent patents in allergy and immunology: Method for treating asthma or allergic disease via anti-Notch4 mAb. Allergy.
[17] Li M,Su B. Up-regulation of NOTCH4 gene expression in bipolar disorder: future studies. Am J Psychiatry. 2013;170 (5):560-1.
[18] Simpson MA,Irving MD,Asilmaz E, et al. Mutations in NOTCH2 cause Hajdu-Cheney syndrome, a disorder of severe and progressive bone loss. Nat Genet. 2011;43 (4):303-5.
[19] McDaniell R,Warthen DM,Sanchez-Lara PA, et al. NOTCH2 mutations cause Alagille syndrome, a heterogeneous disorder of the notch signaling pathway. Am J Hum Genet. 2006;79 (1):169-73.
[20] Brai E,Alina Raio N,Alberi L. Notch1 hallmarks fibrillary depositions in sporadic Alzheimer's disease. Acta Neuropathol Commun. 2016;4 (1):64.
[21] Singh S,Weindorfer C,Nandi A, et al. DLL1-responsive PD-L1 + tumor-associated macrophages promote endocrine resistance in breast cancer. Sci Transl Med. 2025;17 (823):eadr6207.
[22] Xu Y,Qiu Z,Chen J, et al. LINC00460 promotes neuroblastoma tumorigenesis and cisplatin resistance by targeting miR-149-5p/DLL1 axis and activating Notch pathway in vitro and in vivo. Drug Deliv Transl Res. 2024;14 (7):2003-2018.
[23] Xu D,Hu J,Xu S, et al. Dll1/Notch activation accelerates multiple myeloma disease development by promoting CD138+ MM-cell proliferation. Leukemia. 2012;26 (6):1402-5.
[24] Su PL,Chakravarthy K,Furuya N, et al. DLL3-guided therapies in small-cell lung cancer: from antibody-drug conjugate to precision immunotherapy and radioimmunotherapy. Mol Cancer. 2024;23 (1):97.
[25] Rudin CM,Reck M,Johnson ML, et al. Emerging therapies targeting the delta-like ligand 3 (DLL3) in small cell lung cancer. J Hematol Oncol. 2023;16 (1):66.
[26] Owen DH,Giffin MJ,Bailis JM, et al. DLL3: an emerging target in small cell lung cancer. J Hematol Oncol. 2019;12 (1):61.
[27] Hermans BCM,Derks JL,Thunnissen E, et al. DLL3 expression in large cell neuroendocrine carcinoma (LCNEC) and association with molecular subtypes and neuroendocrine profile. Lung Cancer. 2019;138:102-108.
[28] Naseri M,Saeednejad Zanjani L,Vafaei S, et al. Increased cytoplasmic expression of DLL4 is associated with favorable prognosis in colorectal cancer. Future Oncol. 2021;17 (24):3231-3242.
[29] Yan J,Xie Y,Liu Z, et al. DLL4-targeted CAR-T therapy sensitizes neoadjuvant chemotherapy via eliminating cancer stem cells and reshaping immune microenvironment in HER2 + breast cancer. J Immunother Cancer. 2024;12 (11):.
[30] Ding XY,Ding J,Wu K, et al. Cross-talk between endothelial cells and tumor via delta-like ligand 4/Notch/PTEN signaling inhibits lung cancer growth. Oncogene. 2012;31 (23):2899-906.
[31] Wang W,Hu W,Wang Y, et al. Long non-coding RNA UCA1 promotes malignant phenotypes of renal cancer cells by modulating the miR-182-5p/DLL4 axis as a ceRNA. Mol Cancer. 2020;19 (1):18.
[32] Afzalipour R,Abbasi-Dokht T,Sheikh M, et al. The Prediction of DLL4 as a Prognostic Biomarker in Patients with Gastric Cancer Using Anti-DLL4 Nanobody. J Gastrointest Cancer. 2024;55 (3):1380-1387.
[33] Gilbert MA,Keefer-Jacques E,Jadhav T, et al. Functional characterization of 2,832 JAG1 variants supports reclassification for Alagille syndrome and improves guidance for clinical variant interpretation. Am J Hum Genet. 2024;111 (8):1656-1672.
[34] Li G,Liu B,Yang H, et al. Omega-3 polyunsaturated fatty acids alleviate renal fibrosis in chronic kidney disease by reducing macrophage activation and infiltration through the JAG1-NOTCH1/2 pathway. Int Immunopharmacol. 2025;152:114454.
[35] Li C,Wang X,Shi D, et al. RFX5 promotes the progression of triple-negative breast cancer through transcriptional activation of JAG1. Hum Cell. 2025;38 (3):86.
[36] Vaish V,Kim J,Shim M. Jagged-2 (JAG2) enhances tumorigenicity and chemoresistance of colorectal cancer cells. Oncotarget. 2017;8 (32):53262-53275.
[37] Ghoshal P,Nganga AJ,Moran-Giuati J, et al. Loss of the SMRT/NCoR2 corepressor correlates with JAG2 overexpression in multiple myeloma. Cancer Res. 2009;69 (10):4380-7.
[38] Mandula JK,Sierra-Mondragon RA,Chang D, et al. Jagged2 targeting in lung cancer activates anti-tumor immunity via Notch-induced functional reprogramming of tumor-associated macrophages. Immunity. 2024;57 (5):1124-1140.e9.